Curcumin Bioavailability Study
Study Objective
To evaluate the bioavailability of a liposomal curcumin supplement compared to a non-liposomal (standard) curcumin supplement.
Method
The study was a two-group, randomized control trial.
- Group A: Single dose of 250mg liposomal curcumin supplement.
- Group B: Single dose of 250mg standard curcumin supplement.
Participants
Twenty healthy participants were enrolled in the study. They were randomly and evenly assigned to one of the supplementation groups. The participants were between the ages of 20-50 years old, did not have any chronic medical conditions, did not have any presence of acute illness, were within the normal BMI category (18.5-24.9 kg/m2), and did not use any drugs or dietary supplements on a frequent and/or mandatory basis.
Blood Sampling
Participants received their respective dosages of curcumin while fasting. Blood samples were drawn at baseline (before taking the supplement), and at hours 2, 4, and 6 after supplementation to evaluate curcuminoid concentration in the blood.
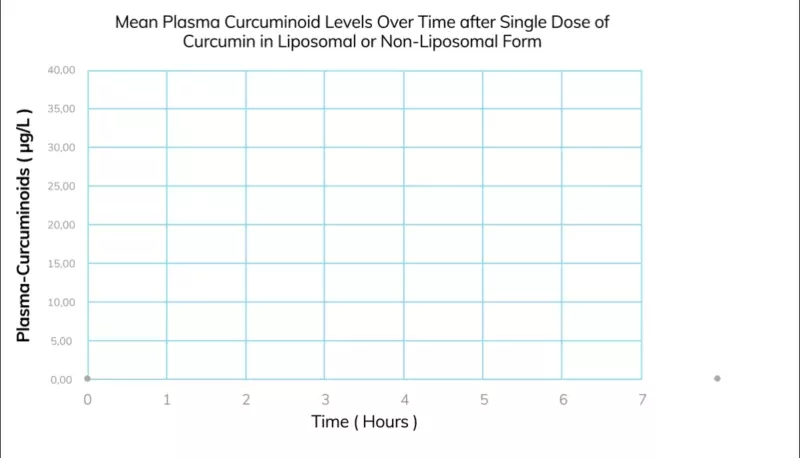
Results
Results of this study show that liposomal curcumin supplementation has higher bioavailability than non-liposomal supplementation.
- Significant differences in curcuminoid values between the two groups were found at hours 2, 4, and 6.
- Liposomal curcumin was found to be 47x more bioavailable than standard curcumin.
Key Takeaways
- Curcumin is a plant extract which has very low bioavailability.
- ActiNovo Liposomal Curcumin has a much higher bioavailability (47x) than a standard curcumin supplement.
- Liposomal curcumin formulas are able to sustain a higher amount of curcuminoids in the blood for a longer period of time than standard curcumin formulas.


